|
Orthologix brings you quarterly information to keep you current on industry news, trends and insights. Orthologix is an orthotics and prosthetics practice delivering evidence-based care throughout the Delaware Valley.

FEATURED PRODUCT
Pediatric Noodle® AFO from Kinetic Research
Dynamic Performance from Heel-Strike to Toe-Off
 The Pediatric Noodle® carbon fiber composite ankle foot orthosis is a lightweight yet extremely durable AFO. It is ideal for users with varying degrees of foot drop who would benefit from a dynamic, energy-storing AFO to improve gait. The flexible footplate and flexible strut facilitate a more natural gait than more rigid carbon fiber AFOs. The Pediatric Noodle® carbon fiber composite ankle foot orthosis is a lightweight yet extremely durable AFO. It is ideal for users with varying degrees of foot drop who would benefit from a dynamic, energy-storing AFO to improve gait. The flexible footplate and flexible strut facilitate a more natural gait than more rigid carbon fiber AFOs.
The Pediatric Noodle AFO provides gait assistance by controlling plantar flexion and reducing energy consumption with dynamic dorsiflexion and improved loading response.
The Noodle AFO is designed to:
- Control drop foot and assist with correct heel strike with gait
- Accommodate plantar flexion tone with variable flexibility footplate
- Facilitate heel-to-toe weight transfer with weight bearing
- Provide flex-assist at toe-off to increase ankle power at push-off
- Provide a swing assist during the swing phase of gait
- Encourage knee flexion with improved ankle/foot biomechanics
- Improve gait efficiency
Indications for the Noodle AFO:
- Simple foot drop with functional plantar flexion strength
- Knee hyperextension control
- Mild Achilles tendon tightness - equinavarus
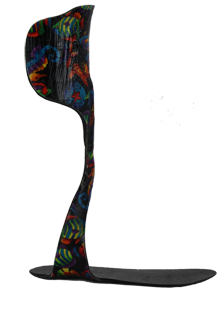 Unique Features of the Noodle AFO: Unique Features of the Noodle AFO:
- Patented carbon fiber composite design that is more flexible than standard carbon fiber AFOs to optimize mobility
- Fusion - Kinetic Research's patent-pending process provides unique product flexibility, which is ideal for patients with mild to moderate foot drop
- Posterior shell design with energy-storing carbon fiber composite dynamic lateral strut and footplate to effectively treat foot drop
Handcrafted Carbon Fiber AFO Brace
The Pediatric Noodle is handcrafted by expert technicians skilled in carbon fiber composite fabrication. This allows for a custom precision fit that optimizes performance and efficacy. The Noodle is available in either Off-the-Shelf, Build-to-Order or Custom-from-Cast models.
Off-the-Shelf Standard Size Models: The footplate can be pre-cut to an exact size at no extra charge.
Build-to-Order Models: Specifications include: cuff height and footplate length variations; strut variation from flexible to extra-stiff; footplate variation from flexible to extra-stiff; colors or user supplied fabric.
Custom-from-Cast Models: For difficult to fit users.
Carbon Fiber Orthopedic Braces – Next Generation Technology
Carbon fiber orthopedic braces from Kinetic Research’s patented products are lighter, stronger and more flexible than standard carbon fiber braces. The key is dynamic flexibility that absorbs and releases kinetic energy. The Noodle flexes and absorbs weight loads well past the point where standard carbon fiber braces fail and break.
Kinetic Research was the first to apply resin sheets and fiber braids in orthotics and prosthetics fabrication. This led to the ReadyPreg® and Ultra-G® lines of materials. Now Kinetic Research has combined ReadyPreg® and Ultra-G® into a patent-pending process called Fusion. The Fusion process is the state-of-the-art in carbon fiber composite technology for orthotics & prosthetics fabrication.
 Variations of the Noodle Variations of the Noodle
The Noodle TA’s rigid anterior shell helps stabilize and extend the knee. Pre-set in two degrees of plantar flexion, the Noodle TA absorbs kinetic energy during the mid through terminal stance phase of gait. That energy is released through the footplate at push-off creating a more natural gait pattern for users that have lost plantar flexion strength. The lateral noodle strut connects with the footplate proximal to the base of the #5 Met Head. This virtually eliminates the uncomfortable contact between the bone and the strut.
The Noodle Plus’s flexible footplate and flexible strut facilitate a more natural gait than more rigid carbon fiber AFO's. Indications for the Noodle Plus are the following:
Mild to moderate knee extension weakness: The Noodle Plus applies a floor reaction force to the upper tibia. This helps with knee control and extension.
Foot/ankle plantar flexion weakness:The Noodle Plus absorbs kinetic energy during the middle and end of the stance phase of the gait cycle. The energy stored is then released at push/toe-off with every step.
Post Tibial Tendonitis Dysfunction (PTTD): The Noodle Plus limits the anterior progression of the tibia which is effective in reducing forces that cause midfoot collapse. It is used in conjunction with foot orthotics to reduce pain and swelling caused by the combination of a tight Achilles tendon, midfoot collapse during the late stages of gait and tendonitis/arthritis associated with the condition.
Partial foot amputation:The Noodle Plus is a lightweight option for mid and distal partial foot prosthetic needs. The energy-storing effect and simulated push-off is ideal for this application. Add a custom insole and shoe filler for a complete solution.
The ValgaNoodle AFO is a new approach to an old orthopedic dilemma. One of the most difficult deformities to address effectively with bracing is ankle and foot varus. In the past, bulky and cumbersome devices would attempt to lock the user’s foot into a static position. The ValgaNoodle AFO uses a dynamic three-point system that moves with the user’s body while applying a constant corrective valgus force. The ValgaNoodle AFO is only available custom-from-cast. The casts are corrected to a more normal foot position and the AFO is fabricated in the improved position. The dynamic properties allow the user to easily don and doff the device. Once applied it creates a constant valgus force designed to improve positioning. The dynamic lateral control coupled with a dynamic footplate leads to more confident and natural gait.
Back to top

PROFILE
 Janet S. Dufek, Ph.D.
Janet S. Dufek, Ph.D.
Dr. Janet Dufek is an associate professor and co-director of the Biomechanics Laboratory at the University of Nevada, Las Vegas. Dr. Dufek’s research focuses on lower extremity function. In collaboration with faculty in engineering, nursing, and physical therapy, her interdisciplinary research addresses issues including child obesity, effectiveness of physical therapy interventions and prosthetic appliances, and aging-related issues. In addition to her academic position, Dr. Dufek is a research scientist with HPW Biomechanics, where she directs research projects related to evaluation of footwear and exercise equipment. Dr. Dufek is a Fellow of the American College of Sports Medicine and the Research Consortium of the American Alliance of Health, Physical Education, Recreation and Dance; the American Society of Biomechanics; and Southwest Regional Chapter of the American College of Sports Medicine.
Her research interests include gait adaptations/balance, lower extremity function, injury prevention mechanisms, variability and human performance, single subject methodology. She has published numerous research papers, a few of which are listed below.
Liebenberg, J., Scharf, J., Forrest, D., Dufek, J.S., Masumoto, K., & Mercer, J.A. Determination of muscle activity during running at reduced body weight. Journal of Sports Sciences. 2011;29:207-214, doi 10.1080/02640414.2010.534806.
Whitley, C.R. & Dufek, J.S. The effect of retro-walking on hamstring flexibility and low back range of motion. International Journal of Exercise Science. 2011;4(3):192-198.
Candela, L., Gutierrez, A.P. Dufek, J.S, Mercer, J.A., & Putney, L.G. Modifying the Diabetes Prevention Program to adolescents in a school setting: A feasibility study. International Scholarly Research Network: Nursing. 2012, Article ID 534085, 9 pages, doi:10/5402/2012/534085.
Dufek, J.S, Currie, R., Gouws, P.L., Candela, L., Gutierrez, A.P., Mercer, J.A., & Putney, L.G. Effects of overweight and obesity on walking characteristics in adolescents. Human Movement Science, 2012;31:897-906, doi:10,1016/j.humov.2011.10.003.
Santo, A.S., Roper, J.L., Dufek, J.S. & Mercer, J.A. Rocker-bottom, profile-type shoes do not increase lower extremity muscle activity and energy cost of walking. Journal of Strength and Conditioning Research. 2012;26(9):2426-2431.
Forrest, D.M., Dufek, J.S. & Mercer, J.A. Impact characteristics of children running in adult sized shoes. Journal of Applied Biomechanics. 2012;28:593-598.
Delgado, T.L., Kubera-Shelton, E., Robb, R.R., Hickman, R., Wallmann, H.W. & Dufek, J.S. Effects of foot strike on low back posture, shock attenuation, and comfort in running. Medicine and Science in Sports and Exercise. Published Ahead of Print, DOI: 10.1249/MSS.0b013e3182781b2c.
Ryan-Wenger, N.A. & Dufek, J.S. An interdisciplinary research model of pediatric patient falls and fall-related injuries. International Journal for Quality in Health Care (in press).
Bates, B.T., James, C.R. & Dufek, J.S. (2003). Single subject analysis. In N. Stergiou (Ed.), Innovative Analyses of Human Movement (pp. 3-28). Champaign, IL: Human Kinetics.
Back to top

LEGISLATIVE NEWS
Fate of Children's Insurance Program Is Called into Question at Senate Hearing
New York Times
WASHINGTON — A Senate hearing on Tuesday set the stage for a coming debate over whether the federal government should continue financing a popular health insurance program for lower-income children who are now eligible for new coverage options under the Affordable Care Act.
The Children’s Health Insurance Program, known as CHIP, has helped cut in half the uninsured rate for children, to about 7 percent in 2013 from 14 percent in 1997, when it was enacted. It provides coverage for about eight million children in families that earn too much to qualify for Medicaid, the government health care program for the poor, but cannot afford private coverage.
The federal government gives matching funds to states to provide the coverage, but that funding is set to end in September 2015, on the assumption that most beneficiaries will be able to get subsidized private coverage through the insurance exchanges created under the health care law. The federal government pays most of the $13 billion annual cost for CHIP.
But advocacy groups are warning that exchange plans will prove unaffordable for many households with children, partly because a quirk in the law prevents families from getting subsidies if a parent is offered “affordable” coverage at work. The law considers premiums on employer-based plans unaffordable if they exceed 9.5 percent of household income, but in what has become known as the “family glitch,” that standard applies only to premium costs for individual plans — not to family plans.
Another concern is that families will face more out-of-pocket costs for exchange plans, and that such plans will offer a narrower set of benefits for children. A recent analysis by the Wakely Consulting Group found that many families would need to spend substantially more out of pocket to get the same care through an exchange plan that they get through CHIP.
So far, only 6 percent of enrollees in the new exchanges are younger than 18, according to the Health Policy Center at the Urban Institute. Far more children are enrolled in Medicaid than either CHIP or the exchange plans, including many who became newly eligible for Medicaid under the Affordable Care Act.
Senator Jay Rockefeller, Democrat of West Virginia, and Representative Henry A. Waxman, Democrat of California, have introduced bills that would extend federal funding for the program through 2019. But the Medicaid and CHIP Payment and Access Commission, an advisory group, has recommended only a two-year extension, saying that would be long enough to address problems like the law’s family glitch.
CHIP has generally had strong bipartisan support, and at Tuesday’s hearing, held by the Senate Finance Committee’s subcommittee on health care, both Democrats and Republicans praised it. But Douglas Holtz-Eakin, a conservative economist and former head of the Congressional Budget Office, pointed out in testimony that the program “now resides in an insurance landscape that is very different than the one it was created in.”
For this reason, he said that “a straight reauthorization is not the best decision” and that “redundancies in coverage should be considered when making funding decisions.”
Mr. Holtz-Eakin added that Congress would have to come up with new resources to pay for an extension of the program, saying, “That’s a problem.”
Senator Michael B. Enzi, Republican of Wyoming, suggested that CHIP at least needed to be whittled down, saying, “We need to focus our efforts here on identifying the core mission of the program.”
Mr. Rockefeller, one of the original architects of the program, who is retiring at the end of this year, said he worried that even with broad support, the program could get caught up in a political fight.
“You can’t always count on something that has been bipartisan to continue to be bipartisan,” he said during the hearing.
Cathy Caldwell, the director of Alabama’s Children’s Health Insurance Program, testified that her state was “already dealing with issues related to the uncertainty of CHIP funding” as it prepared a budget for the 2016 fiscal year.
“We have families already getting stressed,” she said. “They may find coverage through an employer plan or a marketplace plan, but it is my belief that many will not, and they will go back to being uninsured.”
Ronald Pollack, the executive director of Families USA, a liberal-leaning consumer group, said that while he saw “encouraging signs” that the program’s funding would be extended, he worried it could be thwarted by congressional gridlock.
“It’s very possible that it could get caught up in controversial matters extraneous to CHIP,” Mr. Pollack said. “It’s too soon to say.”
A version of this article appears in print on Sept. 17, 2014, on page A18 of the New York edition with the headline: Fate of Children’s Insurance Program Is Called into Question at Senate Hearing.
Frustrated AMA Pitches 'Action Plan' On Digital Records
Kaiser Health News
Saying that electronic health records distract doctors, take time away from care and make physicians less productive, an influential doctors’ group called on vendors and government agencies to work with them to develop better, easier-to-use technology.
The American Medical Association asked the Obama administration to abandon its “all or nothing approach” requiring Medicare providers to go digital or be penalized. The group also wants the government to develop better certification criteria for vendors selling electronic record systems.
It outlined eight areas for overhauling the record systems, with the top priority being to make sure the technology enhances, rather than disrupts patient care. Electronic health records should also promote coordination, enable physicians to delegate to other health care providers and be able to interact with patients’ mobile devices.
Under the 2009 economic stimulus package, Congress authorized $27.4 billion to incentivize doctors to switch from paper to electronic records with the goal of reducing costly errors and duplication and boosting coordination. The law offers doctors who treat Medicare patients up to $63,750 over five years to help pay for the change if they can prove they’re making “meaningful use” of the systems by, say, submitting prescriptions electronically. Those who do not go digital are supposed to have a percentage of their Medicare payments withheld beginning next year. However, the government recently agreed to give certain providers more time and flexibility.
The AMA says that isn’t sufficient to address the problems.
“The meaningful use program and the regulatory structure associated with it initially has been a wonderful impetus to get health systems to adopt [electronic health records],” said Dr. Steven J. Stack, president-elect of the AMA, adding: But the “processes associated with it have become overly prescriptive, rigid and unreasonable and have themselves become a barrier.”
The group said that one of physicians’ chief concerns is that the programs often have clunky menus that require what it called “the collection of time-consuming information of questionable value” and data that is no more than “clutter,” such as lists that do not differentiate between a patient’s current and out-of-date medications.
That information distracts the physician, is often irrelevant to the care of the patient and leads to “excessive clicking and scrolling” on behalf of the doctor, the organization said.
“We do not want to go back to paper records, but today’s current E.H.R. products are immature, costly and not well designed to improve clinical care,” Stack said. “Requiring electronic health records to be all things to all people — regulators, payers, auditors and lawyers — diminishes the ability of the technology to perform the most critical function, helping physicians care for their patients.”
Asked whether they’re seeking delays to the programs penalties beginning next year, Stack said that’s not the group’s goal.
“We would not shy away from accepting delays but have not made that our focus,” he said.
In addition to the criticisms laid out Tuesday, the AMA had joined with the American Hospital Association and 15 other groups to ask the administration to let providers show meaningful use of electronic health records for three months in 2015, as opposed to a full year — arguing the program’s success hinges on the agency offering flexibility.
A spokesman for the Obama administration encouraged doctors to communicate what they need to vendors and others.
“We support the AMA’s efforts to improve EHRs for the end user – the health care providers who use these products to treat their patients,” said Peter Ashkenaz, spokesman for the Office of the National Coordinator for Health Information Technology at the Department of Health & Human Services.
But, he added, “We also know that no software is perfect, and therefore no EHR is perfect.”
Back to top

CONFERENCE
APTA's Combined Sections Meeting, February 4-7, 2015. Indianapolis, Indiana
For more information, go to www.apta.org/CSM
Back to top

Q & A
Please feel free to submit your questions on the Pediatric Noodle® AFO from Kinetic Research as well as any other issue you would like addressed.

Back to top

Educational Workshops and Inservices
Orthologix is available to present educational workshops and inservices for your group or your facility. Email us at info@orthologix.com to schedule.

We hope you have found this issue of the Orthologix E-news informative.
Thank you for your subscription.
Back to top
|